Transdermal Delivery has transformed how medicines and wellness aids reach the body, offering a non-invasive route that bypasses the digestive system and enables steady drug release. Understanding the transdermal patch mechanism helps explain how patches deliver drugs, revealing how adhesion, diffusion, and reservoir design sustain release. This skin absorption transdermal delivery relies on carefully engineered properties such as drug lipophilicity and molecular size to cross the outer skin barrier and enter systemic circulation. Patches provide a flexible alternative to pills by enabling drug delivery through the skin with controlled dosing and reduced gastrointestinal side effects. Safety and efficacy considerations—patch safety and efficacy—remain central as clinicians weigh benefits, risks, and patient tolerability in real-world use.
As an alternative framing, this approach is described as cutaneous or skin-based drug administration, where medicines traverse the skin barriers to reach the bloodstream. Other terms used in the field include dermal patch delivery, transcutaneous administration, and skin permeation strategies that signal related mechanisms and goals. From an LSI perspective, researchers talk about epidermal transport, topical-to-systemic delivery, and patch-mediated diffusion to capture the same core process. Across these terms, the target remains a convenient, reliable route that minimizes digestion, enhances adherence, and supports steady therapeutic exposure.
1) Transdermal Delivery: Core Principles, Patch Mechanism, and Clinical Relevance
Transdermal Delivery represents a non-invasive route for medications and wellness aids that bypasses the digestive system and liver metabolism. The patch mechanism relies on diffusion, partitioning, and controlled release to move the drug from a reservoir or polymer matrix through the skin and into systemic circulation. By design, the drug must be sufficiently lipophilic to traverse the skin’s lipid-rich barrier while still achieving a therapeutic concentration once in the bloodstream. Understanding the basics of how patches deliver drugs helps explain their potential advantages and limitations in therapy, prevention, and symptom management.
This overview emphasizes the practical outcomes of transdermal delivery: steady drug flux, improved patient convenience, and the possibility of reducing peaks and troughs in blood levels. Clinicians and patients can evaluate when transdermal therapy is appropriate, recognizing that patch performance depends on formulation, skin condition, and adherence to wear-time guidelines. The discussion also sets the stage for comparing transdermal approaches with oral, injectable, or nasal delivery methods in various therapeutic contexts.
2) How Patches Deliver Drugs: Diffusion, Reservoirs, and Adhesion
At the heart of the delivery process is the sequence of diffusion, partitioning, and sustained diffusion from the patch into the skin. The patch may use a reservoir or a polymer matrix to modulate how quickly the drug moves toward the stratum corneum, then into viable skin layers and ultimately into the bloodstream. This description aligns with the concept of the transdermal patch mechanism and illustrates how design choices influence the rate and consistency of drug release.
Adhesion, reservoir design, and the drug’s physicochemical properties collaborate to determine actual delivery. Good adhesion maintains contact without causing irritation, while reservoir systems or matrix matrices control the amount released over time. Real-world factors such as placement, activity, and skin hydration can shift the delivered dose, highlighting the importance of fit, site rotation, and patient education to maximize predictable outcomes.
3) Skin Absorption Transdermal Delivery: The Skin Barrier and Permeation
The skin acts as both a barrier and a conduit for transdermal delivery. The stratum corneum, composed of lipids and dead cells, is the primary obstacle that soluble and lipophilic molecules must cross. Drug properties such as lipophilicity, molecular size, and solubility influence skin absorption transdermal delivery, with permeation governed by interactions at the lipid-rich barrier and within the viable epidermis and dermis.
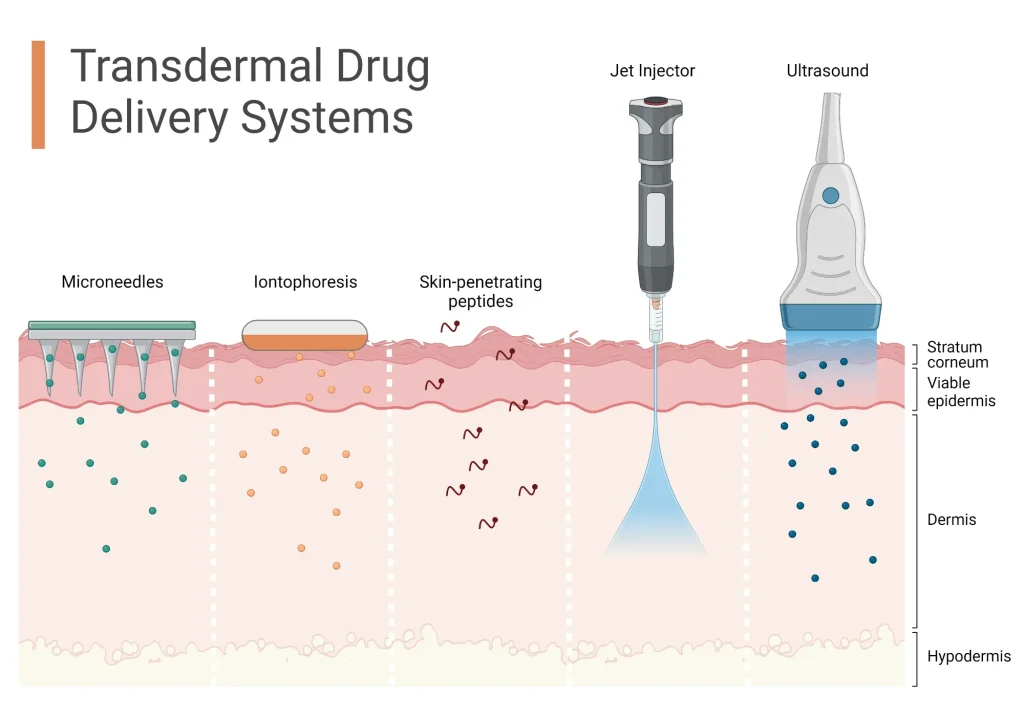
To overcome barrier limits, formulation strategies may employ chemical enhancers, microneedle accents, ultrasound, or other techniques that temporarily loosen skin lipids or create microchannels. Each approach aims to increase bioavailability while balancing safety and tolerability. Understanding these permeation pathways helps explain why some compounds are well-suited to patches and others are not, as well as how skin condition and site selection impact absorption.
4) Drug Delivery Through the Skin: Formulation Strategies for Stable Release
Formulation strategies determine how a drug is held and released from the patch. Matrix patches embed the drug in a polymer that gradually releases it, while reservoir patches contain a drug solution in a gel-like layer separated by a membrane. Some designs incorporate chemical enhancers or novel materials to accelerate diffusion, and hybrid approaches may combine microneedles or iontophoresis to expand the range of deliverable therapies.
These strategies aim to maximize bioavailability while minimizing irritation and inter-patient variability. The choice between continuous release versus step changes in exposure reflects clinical goals and patient lifestyle. Dosing regimens, wear time, and patch size all influence the actual amount delivered, making formulation decisions central to achieving reliable therapeutic outcomes.
5) Patch Safety and Efficacy: Minimizing Irritation and Optimizing Outcomes
Patch safety and efficacy are guided by factors such as skin tolerance, potential irritation, contact dermatitis, and the risk of dose dumping if the patch is damaged or improperly worn. The adhesive layer must balance secure attachment with skin friendliness, and occlusion can alter hydration and drug flux. Regulators also emphasize stable dosing, consistent delivery, and clear labeling to support safe switching between patch types or discontinuation.
Clinicians monitor pharmacokinetic goals alongside patient-specific considerations—age, skin condition, and comorbidities can influence response. Education on site rotation, wear duration, and disposal, along with follow-up assessments after initiating a patch, helps ensure therapy remains within the therapeutic window. In practice, a careful safety profile and evidence of consistent efficacy underpin the broader acceptability of transdermal therapies.
6) Future Directions in Transdermal Patch Technology: Microneedles, Iontophoresis, and Smart Delivery
Advances in transdermal patch technology include microneedle-assisted systems that temporarily bypass the stratum corneum, enabling delivery of larger molecules or vaccines with improved control. Iontophoresis uses a small electrical current to drive charged drugs across the skin, modulating flux and expanding the range of deliverable therapies. These innovations are reshaping how patches achieve higher bioavailability while maintaining a patient-friendly profile.
The future also envisions safer chemical enhancers, responsive patch designs, and integration with wearables or digital health monitoring. As researchers refine permeation strategies and ensure long-term safety, transdermal patches may become viable options for increasingly diverse indications—from chronic disease management to preventive therapies—further blurring the line between drug delivery through the skin and continuous, user-centered care.
Frequently Asked Questions
What is Transdermal Delivery and how does the transdermal patch mechanism work to deliver drugs?
Transdermal Delivery is a non-invasive route that moves a drug from a patch into the bloodstream through the skin. The transdermal patch mechanism relies on diffusion from a reservoir or polymer matrix, across the stratum corneum, into viable skin, and into systemic circulation, delivering a steady flux rather than a bolus dose. Successful delivery requires balancing lipophilicity, molecular size, and patch design to sustain therapeutic levels while minimizing irritation.
How do patches deliver drugs through the skin to achieve systemic exposure?
In Transdermal Delivery, patches deliver drugs by releasing them from a reservoir or drug-in-adhesive layer. The drug diffuses through the stratum corneum into the epidermis and dermis, then enters the bloodstream for systemic exposure. The design aims for continuous release to maintain steady plasma levels and reduce peaks that can cause side effects.
Which factors affect patch safety and efficacy in Transdermal Delivery?
Patch safety and efficacy depend on drug properties (molecular weight, lipophilicity, solubility), skin condition (hydration, irritation), placement site, and user factors like age and activity. The dose must balance achieving a therapeutic effect with minimizing adverse events, and skin reactions or irritation are important considerations in evaluating a patch’s safety and efficacy.
What are common patch types and their real-world applications within Transdermal Delivery?
Common patch types include nicotine replacement therapy patches, pain-management patches, and hormone patches (estrogen, testosterone), illustrating how drug delivery through the skin can provide steady exposure without injections or oral dosing. Antiemetic patches like scopolamine also demonstrate utility in motion sickness, highlighting the range of clinical uses and the importance of patch safety and efficacy.
What are the main limitations of drug delivery through the skin in transdermal systems?
A key limitation is that the skin acts as a barrier to many drugs, making delivery through the skin challenging for large, charged, or highly hydrophilic molecules. Variability in absorption due to skin condition, site, temperature, and activity, plus potential patch irritation, can affect outcomes. Accurate dosing and regulatory labeling are essential for safe, effective use.
What future directions are shaping Transdermal Delivery, and how might innovations like microneedles and iontophoresis enhance the transdermal patch mechanism?
Future directions in Transdermal Delivery include microneedle-assisted patches that temporarily bypass the stratum corneum, and iontophoresis to modulate drug flux across the skin. Chemical enhancers and advanced patch designs aim to expand the range of deliverable drugs while maintaining safety and efficacy, advancing the transdermal patch mechanism and overall skin absorption transdermal delivery.
| Aspect | Key Points from Base Content | Notes / Relevance |
|---|---|---|
| Overview | Transdermal Delivery is a non-invasive route that bypasses the digestive system, enabling steady drug release over hours or days and improving convenience and adherence. | Intro-level summary from base content. |
| Mechanism | Delivery relies on diffusion, partitioning, and controlled release; drug diffuses from patch through stratum corneum to viable skin layers and into systemic circulation; requires adequate lipophilicity and ability to reach therapeutic blood levels. | Patch design uses adhesion, diffusion, and reservoir strategies for sustained release. |
| Skin barrier and patch design | Stratum corneum is the primary obstacle; patch technologies optimize properties (lipophilicity, molecular size) and use adhesive matrices, drug-in-adhesive layers, or reservoirs to create a steady flux. | Steady flux maintains stable levels with fewer peaks. |
| Formulation strategies | Matrix patches embed drug in polymer for slow release; reservoir patches store drug in a gel-like layer diffusing through a membrane; enhancers, ultrasound, microneedles, or iontophoresis can boost flux. | Aims to maximize bioavailability while minimizing irritation and inter-user variability. |
| Factors affecting performance | Drug properties (molecular weight, lipophilicity, solubility) govern penetration; appropriate drugs are favored; unsuitable drugs may require special design. | Skin condition, hydration vs. dryness; occlusion; site-specific factors (arm, chest, back); activity, temperature, sweating; dosing vs. design; placement/skin prep affect actual dose. |
| Patch types & applications | Nicotine patches, pain-management patches, hormone patches (estrogen, testosterone), antiemetic patches (scopolamine); diverse applications beyond nicotine replacement. | Different therapeutic domains with safety and efficacy considerations. |
| Advantages | Ease of use, improved adherence, avoidance of first-pass metabolism, and ability to maintain steady plasma concentrations; can reduce GI side effects for chronic therapy. | Not all drugs are suitable; potential skin irritation; variability; regulatory considerations. |
| Limitations | Not all drugs permeate well; large, charged, or highly hydrophilic molecules may be unsuitable; irritation and variability are concerns. | Requires careful patient selection and monitoring. |
| Future directions | Microneedle-assisted patches, iontophoresis, chemical enhancers are advancing to broaden drug range and dosages. | Ongoing research to improve safety and comfort. |
| Practical considerations | Choosing patches involves efficacy, convenience, and skin tolerance; rotate sites, wear time, and disposal; monitor therapy and adjust dosing as needed. | Real-world use considerations for clinicians and patients. |
Summary
Transdermal Delivery represents a compelling approach to drug administration that blends skin science with practical design to deliver consistent, patient-friendly therapy. The transdermal patch mechanism is a careful balance of drug properties, patch formulation, and skin biology, optimized to achieve a steady flux from patch to bloodstream. By understanding how patches deliver drugs and the factors that influence absorption, clinicians and patients can select appropriate patch technologies for a range of conditions, from nicotine dependence to chronic pain and hormonal therapy. While challenges remain—such as variability in skin response and the need for suitable molecular characteristics—the future of transdermal delivery holds promise with innovations like microneedles and iontophoresis. In sum, transdermal delivery is not just a delivery method; it is a sophisticated interplay of material science, pharmacology, and dermatology designed to improve health outcomes, patient experience, and everyday life.